Org. Synth. 1972, 52, 100
DOI: 10.15227/orgsyn.052.0100
3β-ACETOXY-5α-CYANOCHOLESTAN-7-ONE
[Cholestane-5-carbonitrile, 3-(acetyloxy)-7-oxo-, (3β, 5α)-]
Submitted by W. Nagata and M. Yoshioka
1.
Checked by Robert E. Ireland, Robert Czarny, and Conrad J. Kowalski.
1. Procedure
Caution! This preparation should be carried out in a good hood. Also, great care should be taken in handling neat triethylaluminum because it is pyrophoric—that is, it ignites spontaneously upon contact with air (Note 2).
Benzene has been identified as a carcinogen; OSHA has issued emergency standards on its use. All procedures involving benzene should be carried out in a well-ventilated hood, and glove protection is required.
A dry, 50-ml., three-necked, round-bottomed flask equipped with a gas-inlet tube for nitrogen, a magnetic stirring bar, and a serum stopper for the introduction of reagents is flushed with nitrogen, stoppered with a glass stopper, charged with 17 ml. of anhydrous tetrahydrofuran (Note 1), then immersed in an ice bath. Stirring is begun and 3.3 g. (3.9 ml., 0.028 mole) of triethylaluminum is introduced into the flask with a dry hypodermic syringe (Note 2), (Note 3). After 5–10 minutes, 4.8 ml. of a 3.57 M solution of hydrogen cyanide (0.017 mole) in anhydrous tetrahydrofuran (Note 4) is added with a dry hypodermic syringe. The stirring is continued for about 5–10 minutes.
A dry, 100-ml., three-necked, round-bottomed flask equipped with a gas-inlet tube for nitrogen, a magnetic stirring bar, and a serum stopper, as described above, is flushed with nitrogen. While the flask is being flushed, 2.50 g. (0.00565 mole) of 3β-acetoxycholest-5-en-7-one (Note 5) and 0.0521 ml. (0.00289 mole) of water (Note 6) are added to the reaction flask. The flask is stoppered with a glass stopper and charged with 17 ml. of anhydrous tetrahydrofuran. After the starting material has dissolved, the cold triethylaluminum-hydrogen cyanide solution is transferred to the reaction flask with a dry hypodermic syringe. The resulting pale yellow solution is stirred at room temperature under a positive nitrogen pressure. After 3 hours, a solution of 0.044 ml. (0.0024 mole) of water in 0.87 ml. of anhydrous tetrahydrofuran is added, and the solution is allowed to stir for an additional 4 hours.
The reaction mixture is poured slowly into a vigorously stirred solution of 28 ml. (0.28 mole) of concentrated hydrochloric acid and 350 ml. of ice water in a 1-l., three-necked, round-bottomed flask fitted with an efficient stirrer and immersed in an ice bath (Note 7), (Note 8). The mixture is stirred for 20 minutes with ice cooling and extracted with three 200-ml. portions of a 3:1 (v/v) mixture of diethyl ether and dichloromethane. The extracts are washed with three 200-ml. portions of aqueous 2 M sodium hydroxide, two 200-ml. portions of water, and one 200-ml. portion of saturated aqueous sodium chloride, dried over anhydrous sodium sulfate, and evaporated under reduced pressure (Note 9) and (Note 10). The crystalline residue, weighing 2.70 g., is recrystallized by dissolving it in 7.5–8 ml. of hot (almost boiling) benzene and adding 25 ml. of n-pentane (distilled) to the hot solution (Note 11). 3β-Acetoxy-5α-cyanocholestan-7-one is obtained as white crystals, m.p. 192.5–193.5°; the yield is 2.27–2.41 g. (86–91%). A second crop (50–170 mg.) can be obtained, m.p. 188.5–190° (Note 12); the total yield is 92–93% (Note 13) and (Note 14).
2. Notes
1.
Prior to use the
tetrahydrofuran was distilled from
lithium aluminum hydride into a dry flask flushed with
nitrogen and sealed with a serum stopper.
2.
Caution! Triethylaluminum is pyrophoric. Use safety glasses, gloves, and an apron. Use dry sand to extinguish fires. The submitters note that a description of the properties and handling procedures for
triethylaluminum are available from the Ethyl Corporation, Louisiana. The checkers used
triethylaluminum in lecture bottles from Alpha Inorganics, Inc., and suggest the handling procedure described below. Since this procedure was submitted and checked,
standardized solutions of triethylaluminum in various hydrocarbon solvents, which may be substituted for pure triethylaluminum, have become available from Texas Alkyls, Inc., a division of Stauffer Chemical Company.
a.
Checkers handling procedure.
Figures 1 and
2 suggest the equipment to be used, and how to assemble it. The hose end fitting is connected to the stopcock with a piece of
Teflon-lined tubing and fastened with
copper wire. The stopcock should be well greased and held secure with a
taut rubber band since it does have a tendency to pop out. The
triethylaluminum may now be removed from the lecture bottle by the following procedure. A
dry, three-necked flask equipped with serum stopper and gas-inlet tube is flushed thoroughly with nitrogen from a nitrogen bubbler. The triethylaluminum transfer apparatus is put into the open neck of the flask, the joint being well greased (the gas-inlet tube joint should also be greased). With the stopcock of the transfer apparatus
open and well secured as suggested above, the tank valve is opened (usually one or two full turns) with a wrench. The flow of
triethylaluminum may now be adjusted with the stopcock. When one obtains as much
triethylaluminum as desired, the tank valve is closed, the transfer apparatus is allowed to drain, and the stopcock is closed. The transfer apparatus is removed from the flask which is quickly stoppered with a
glass stopper while the flask is flushed with
nitrogen. This flask of
triethylaluminum can be stored in this manner for many weeks since this reagent is quite stable. The transfer apparatus, which may still contain some
triethylaluminum, should be carefully removed, rinsed quickly with
acetone, and cleaned with
dilute hydrochloric acid.
Figure 1. Apparatus for collecting triethylaluminum from a lecture bottle.
Figure 2. Detailed construction of the stopcock for the apparatus shown in
Figure 1.
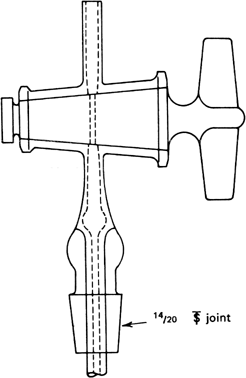
b.
Submitters handling procedure. The submitters suggest another handling procedure especially useful for removing
triethylaluminum from a
lecture bottle having a clogged valve outlet.
Figure 3 shows the apparatus and how to transfer the material. With the cylinder clamped in upright position, remove the valve unit so that only the bottle remains. Quickly attach the adaptor to the opening of the bottle and apply a slow stream of
nitrogen. Transfer the
triethylaluminum into
dry, nitrogen-flushed, 50-ml. ampoules using a
100- or 200-ml. Luer-lock hypodermic syringe with a needle, 43 cm. long and 2 mm. in diameter. Sweep the opening of the ampoule with
nitrogen during the transfer. The syringe should be slightly greased, and the ampoules should be strong with a long, thick stem so that they can be resealed. The ampoules are sealed as soon as possible. The use of rubber caps is effective for temporary protection of the ampoules from air. The material in a 50-ml. ampoule can be divided in smaller ampoules using the apparatus shown in
Figure 4, which can be used also for transferring
triethylaluminum from an ampoule to a reaction flask.
Figure 3. Apparatus for removing triethylaluminum from the lecture bottle.
Figure 4. Apparatus for collecting aliquots of triethylaluminum.
This procedure was not tested by the checkers. It has the advantage that one does not have to contend with a clogged lecture bottle, which in the checkers experience is best discarded. It has the disadvantage that one must handle large syringe-fulls of triethylaluminum and make several transfers without exposing the liquid to air. Caution must be exercised with either procedure.
3.
Neat
triethylaluminum may be replaced with a
10–25% stock solution of it in anhydrous tetrahydrofuran, decreasing the amount of solvent in the reaction flask. The stock solution is prepared using a
graduated flask to measure the volume of the
triethylaluminum and solvent, added appropriately. The stock solution is very stable and not pyrophoric.
4.
The submitters have prepared
hydrogen cyanide as directed in
Org. Synth., Coll. Vol. 1, 314, (1944). The checkers used a similar procedure described by Brauer.
2 The
hydrogen cyanide was collected in a tared flask and diluted with anhydrous
tetrahydrofuran to a previously marked volume. The flask was capped with two serum stoppers (the second put on in an inverted position) to ensure against leakage and stored in a freezer. Solutions such as these seem to be stable for several months when kept cold.
5.
Checkers obtained the steroid from K & K Laboratories.
6.
A small amount of water has been found to accelerate the reaction. In the absence of water, the reaction was about 80% complete after 7.5 hours. Despite the rate acceleration by water, the reaction mixture should be protected from moisture, because a larger amount of water than that specified retards the reaction, owing to decomposition of the
triethylaluminum.
7.
This treatment is an exothermic reaction with evolution of
gaseous ethane. The reaction mixture should be added in a slow stream, with good stirring, at such a rate that the content in the flask does not overflow. When the ice has melted, additional ice should be added.
8.
The reaction mixture remaining on the wall of the reaction flask is treated with a small amount of cold, dilute
hydrochloric acid and combined with the extraction mixture.
9.
Acid treatment followed by alkaline washing of the extracts prevents possible hydrolysis of the acetoxyl and the cyano groups.
10.
The submitters have performed the extraction and washing in a countercurrent manner using three
2-l. separatory funnels.
11.
The recrystallization was carried out in a
40-ml. centrifuge tube. The recrystallization mixture, after cooling to room temperature, was cooled in a freezer and washed twice with 20-ml. portions of cold recrystallization solvent.
12.
When the second crop is contaminated with a polar material (α-cyanohydrin of the product) as evidenced by a lower melting point (m.p.
160–170°) and TLC (Kiesel Gel GF,
benzene-ethyl acetate, 4:1,
Rf = 0.2), the residue from the mother liquor must be treated again with alkali before crystallization.
13.
This procedure is applicable to smaller or larger scale preparations with some modification. In a smaller scale experiment, the submitters suggest using a stock solution of
triethylaluminum (Note 3). In a larger scale preparation, to avoid using large syringes, it is possible to run the
triethylaluminum into a
dry, graduated, pressure-equalizing dropping funnel, in which case the
triethylaluminum-hydrogen cyanide solution may be added to the reaction flask through a funnel instead of using a syringe.
14.
The submitters used
5.76 g. of 3β-acetoxycholest-5-en-7-one and obtained
5.65 g. (
92%) of the cyano ketone.
3. Discussion
3β-Hydroxy-5α-cyanocholestan-7-one has been prepared in
43% yield by the action of
potassium cyanide and
ammonium chloride3 on
7-ketocholesterol at 100° for 33 hours.
4 The present method was developed by the submitters.
4
This process is superior to classical hydrocyanation methods using an alkali metal cyanide
5 and to the improved method using
potassium cyanide and
ammonium chloride3 with respect to reactivity, stereospecificity, and absence of side reactions. Also, the process is applicable to conjugate hydrocyanation of various α,β-unsaturated carbonyl and imino compounds,
4,6 cyanohydrin formation from less-reactive ketones,
4 and cleavage of epoxides to produce β-cyanohydrins,
7 the reaction conditions being varied depending on the substrate to be used. The present procedure is typical of hydrocyanation procedures using other organoaluminum compounds.
Cyanotrimethylsilane reacts with α,β-unsaturated ketones in the presence of
triethylaluminum to give trimethylsilyl enol ethers of β-cyano ketones.
8
Appendix
Chemical Abstracts Nomenclature (Collective Index Number);
(Registry Number)
hydrochloric acid (7647-01-0)
Benzene (71-43-2)
diethyl ether (60-29-7)
ammonium chloride (12125-02-9)
sodium hydroxide (1310-73-2)
sodium chloride (7647-14-5)
hydrogen cyanide (74-90-8)
sodium sulfate (7757-82-6)
nitrogen (7727-37-9)
potassium cyanide (151-50-8)
acetone (67-64-1)
n-PENTANE (109-66-0)
dichloromethane (75-09-2)
ethane (74-84-0)
Tetrahydrofuran (109-99-9)
lithium aluminum hydride (16853-85-3)
triethylaluminum (97-93-8)
3β-ACETOXY-5α-CYANOCHOLESTAN-7-ONE,
Cholestane-5-carbonitrile, 3-(acetyloxy)-7-oxo-, (3β, 5α)- (2827-02-3)
3β-acetoxycholest-5-en-7-one
triethylaluminum-hydrogen cyanide
3β-Hydroxy-5α-cyanocholestan-7-one
7-ketocholesterol
Cyanotrimethylsilane (7677-24-9)
Copyright © 1921-, Organic Syntheses, Inc. All Rights Reserved