Org. Synth. 1984, 62, 58
DOI: 10.15227/orgsyn.062.0058
SILYLATION OF 2-METHYL-2-PROPEN-1-OL DIANION: 2-(HYDROXYMETHYL)ALLYLTRIMETHYLSILANE
Submitted by Barry M. Trost, Dominic M. T. Chan, and Thomas N. Nanninga
1.
Checked by Paul R. Jenkins and Ian Fleming.
1. Procedure
Caution! Part A should be carried out in an efficient hood, since the reagents are noxious.
A. 2-(Trimethylsiloxymethyl)allyltrimethylsilane. An oven-dried (Note 1) 2-L, three-necked, round-bottomed flask is equipped with an airtight mechanical stirrer (Note 2), a 500-mL pressure-equalizing dropping funnel (Note 3), and a reflux condenser. The top of the condenser is connected to a three-way stopcock with one branch connected to a nitrogen source and the other to a variable pressure oil pump with a dry ice trap (Note 4). The apparatus is flamed dry under a steady stream of nitrogen. The flask is charged with 836 mL (1.07 mol) of a 1.28 M solution of butyllithium in hexane (Note 5). The bulk of the hexane is removed at reduced pressure with stirring until a thick oil is obtained (Note 6). The system is carefully recharged with nitrogen. Alternatively, the use of 107 mL of 10 M butyllithium in hexanes is more convenient and gives similar yields (Note 7). The butyllithium is then cooled in an ice bath and 500 mL of anhydrous ether is added (Note 8), followed by 160 mL of tetramethylethylenediamine (Note 9). The mixture is stirred for a few minutes and 34 mL, 29.14 g (0.404 mol) of 2-methyl-2-propen-1-ol (Note 10) is added dropwise via a syringe over 22 min (Note 11). An immediate, vigorous reaction occurs and the lithium alkoxide precipitates as a white solid. Approximately 350 mL of tetrahydrofuran (Note 12) is added and the resultant slightly cloudy yellow solution is allowed to warm to room temperature over ca. 4 hr (Note 13). The reaction is stirred for 39 hr (Note 14), at which time the dianion separates as a dark-red gummy material from the deep orange solution. The mixture is cooled to ca. −30°C (Note 15) and 230 mL (1.81 mol) of chlorotrimethylsilane (Note 16) is added all at once over ca. 20 sec. The reaction turns milky white (Note 17). After 5 min, the dry ice bath is removed and the mixture is stirred for a further 15 min at room temperature. The reaction mixture is added in two portions with swirling to 1.5 L of ether in two 2-L conical flasks, after which 1 L of saturated aqueous sodium bicarbonate is added very carefully to destroy excess chlorotrimethylsilane (Note 18). The two layers are separated and the aqueous phase is extracted with a further 1.5 L of ether. The combined organic layer is then washed with 1 L of water, two 1-L portions of saturated aqueous copper sulfate solution, and 400 mL of water. The solution is dried over anhydrous potassium carbonate and the solvent is removed by atmospheric distillation (Note 19). Careful distillation of the residual oil through a 27-cm Vigreux column at reduced pressure gives a forerun of 4.25 g, bp 29–57°C (4 mm), and 45.8 g (52%) of 2-(trimethylsiloxy)allyltrimethylsilane as a colorless liquid, bp 57–59°C (4 mm) (Note 20).
B. 2-(Hydroxymethyl)allyltrimethylsilane. A 500-mL round-bottomed flask equipped with a magnetic stirring bar is charged with 21.10 g (0.0975 mol) of 2-(trimethylsiloxymethyl)allyltrimethylsilane in 170 mL of tetrahydrofuran (Note 12) and 44 mL of ca. 1 N aqueous sulfuric acid (Note 21). The resultant two-phase mixture is then stirred vigorously for 1.5 hr at room temperature. Solid anhydrous potassium carbonate is added carefully until bubbling subsides. The layers are separated and the aqueous layer is extracted with 100 mL of ether. The combined organic layers are dried over anhydrous potassium carbonate and distilled at atmospheric pressure to remove the solvents (Note 19). The remaining liquid is distilled at reduced pressure to give a forerun, 0.4 g, bp 22–54°C (4 mm), and 10.95 g (78%) of 2-(hydroxymethyl)allyltrimethylsilane as a colorless liquid, bp 54–56°C (2 mm) (Note 22).
2. Notes
1.
All glassware was dried in an oven at over 100°C overnight.
2.
The use of a magnetic stirrer is not advisable since the formation of the gum-like dianion prevents efficient stirring. A
mechanical stirrer with a ground-glass shaft bearing lubricated with mineral oil is recommended.
3.
The funnel is capped with a
rubber septum. For ease of operation, volume markings, corresponding to the amounts of reagents to be added, are put on the addition funnel.
4.
The function of the
trap is to condense the
hexane from the
butyllithium solution. The checkers used a
1-L three-necked flask fitted with a short
delivery tube (a
quick-fit air-bleed tube was used), stopper, and rubber tubing connection. The submitters used a
water aspirator and a
1-L filter flask with a drying tower between.
5.
Butyllithium in hexane was purchased by the checkers from Pfizer Chemicals Ltd., UK, and manufactured by the Lithium Corporation of America. It was titrated using the double titration method with
dibromoethane and transferred to the addition funnel using a cannula. The submitters used a 1.58
M solution from the Foote Mineral Company; they found that the yield of product was reduced to ca. 42% when only 2 equiv of the
lithium reagent was used.
6.
One should try to remove as much
hexane as possible from the
butyllithium solution (i.e. greater than 90%) because the purity of the product depends on the polarity of the reaction medium. A warm-
water bath was used to facilitate solvent removal. The checkers used a variable pressure oil pump with the vacuum adjusted to ca. 10–20 mm.
7.
Butyllithium as a 10 M solution in hexanes is supplied by Aldrich Chemical Company, Inc. The procedure for its use in place of solutions of lower concentration is identical to that in the text except for deletion of the solvent evaporation step. As 10
M butyllithium is quite viscous and pyrophoric it should be handled with large-bore needles.
8.
Ether was distilled from
sodium ketyl of benzophenone. The dissolution of
butyllithium in ether was slightly exothermic.
9.
Tetramethylethylenediamine was obtained from Aldrich Chemical Company and distilled from
calcium hydride before use.
10.
2-Methyl-2-propen-1-ol, purchased from Aldrich Chemical Company, was distilled from
anhydrous potassium carbonate. It was added directly to the
butyllithium solution using a long needle. The checkers quickly replaced the
pressure-equalizing dropping funnel with a serum cap to carry out this addition. The funnel was fitted to a small dry flask to prevent the introduction of moisture during the addition period and replaced on the reaction flask immediately afterward.
11.
The reaction of the alcohol with
butyllithium is quite vigorous with evolution of
butane.
12.
Tetrahydrofuran was distilled from
sodiumketyl of benzophenone.
13.
The checkers renewed the
ice bath when additions were complete and allowed the flask to remain in the
ice bath without addition of fresh ice.
14.
Dianion formation appears to be essentially complete within 24 hr. However, a reaction time of 36 hr is recommended by the submitters to ensure complete reaction.
15.
An extremely violent reaction is observed if the dianion is quenched above 0°C, with ether boiling off at an uncontrollable rate. The submitters observed that if the
chlorotrimethylsilane addition is performed at a lower temperature, the reaction temperature will remain below that of the boiling point of ether. A
dry-ice bath made up of 80 : 20 (v/v)
ethanol : water was used; the checkers measured a bath temperature of −55°C and kept the reaction in the bath for 15 min before adding
chlorotrimethylsilane.
16.
Chlorotrimethylsilane was distilled from
tributylamine before use. Both of these reagents were obtained from the Aldrich Chemical Company.
17.
The submitters observed the appearance of a brown color at this point. The checkers obtained a brown color only after the reaction mixture was added to
ether. In a run at half scale the reaction mixture remained milky white for 35 min and turned brown only when
ether (500 mL) was added to it.
18.
The submitters observed more precipitate on dilution with ether and recommended that the aqueous workup be performed in a
hood.
19.
The submitters distilled most of the solvent using a bath temperature increasing up to 100°C. The checkers used a
rotary evaporator with a hot
water bath.
20.
A variable pressure oil pump was used in this distillation. Approximately 10 g of a volatile component, consisting mostly of
hexamethyldisiloxane, was obtained at room temperature (15 mm) before the foreun. The forerun contained the desired product and mineral oil from the
butyllithium solution. The pot residue was about 5 g. The submitters find the disilyl compound thus obtained is contaminated with a trace amount of
mineral oil and
4–6% of a vinylsilane, probably
2-methyl-1-trimethylsiloxy-3-trimethylsilyl-2-propene. This impurity becomes quite significant if the reaction medium is less polar than the one described (e.g., too much
hexane from
butyllithium is allowed to remain behind). The spectral properties of the desired product determined by the checkers are as follows: IR (neat) cm
−1: 2955, 1643, 1636, 1250, 1085, 885–830;
1H NMR (chloroform-
d, 90 MHz) δ: 0.03 [s, 9 H, CH
2Si(CH
3)
3], 0.14 [s, 9 H, OSi(CH
3)
3], 1.50 [broad s, 2 H, CH
2-Si(CH
3)
3], 3.93 [broad s, 2 H, CH
2-OSi(CH
3)
3], 4.62 (m, 1 H, vinyl H), 4.92 (m, 1 H, vinyl H). The checkers observed small NMR peaks assigned to mineral oil at δ 0.9 and 1.28 and peaks assigned to 2-methyl-1-trimethylsiloxy-3-trimethylsilyl-2-propene at δ 1.87 and 4.1. When the reaction was carried out at half scale the quantity of the latter impurity was not measurable from the NMR integral; however, a run at full scale gave about 10% of the impurity as estimated from the NMR integral. The product from the run at half scale had bp
56–57°C (2 mm); the submitters, bp
65°C (5.5 mm).
21.
The acid solution was prepared by adding
13.5 mL of concentrated sulfuric acid to 500 mL of distilled water.
22.
A
variable-pressure pump is used for the distillation. The forerun consisted of mineral oil contaminant and product. The
allylic alcohol is not very stable at room temperature but can be maintained indefinitely in the
refrigerator at 0 to −6°C. The spectral properties of the alcohol were determined by the checkers as follows: IR (neat) cm
−1: 3600–3100, 2950, 1643, 1637, 1247, 1050, 885–830;
1H NMR (chloroform-
d), 90 MHz) δ: 0.02 [s, 9 H, CH
2Si(CH
3)
3], 1.51 [s, 2 H, CH
2-Si(CH
3)
3], 2.16 (broad s, 1 H, OH), 3.92 (broad s, 2 H, CH
2-OH), 4.62 (m, 1 H, vinyl H), 4.98 (m, 1 H, vinyl H). The checkers observed small NMR peaks assigned to mineral oil at δ 0.82 and 1.51 and peaks assigned to 2-methyl-1-trimethylsiloxy-3-trimethylsilyl-2-propene at δ 0.10, 1.87, and 4.1. When the reaction was carried out at half scale the quantity of the latter impurity was reduced; the product from the run at half scale had bp
54–55°C (3 mm); submitters, bp
53–54°C (1.6 mm).
3. Discussion
Compound
1,
2-(hydroxymethyl)allyltrimethylsilane, represents a conjunctive reagent that can be considered as the equivalent of zwitterion
2, possessing a nucleophilic
allylanion synthon and an electrophilic allyl cation synthon in the same molecule. It has been employed in a three-carbon condensative ring-expansion reaction,
2 a [3 + 2] annulation with cyclic enones,
3,4 and the total synthesis of coriolin.
5 Acetylation of the
allylic alcohol gives
2-(acetoxymethyl)allyltrimethylsilane, which undergoes
palladium(0) catalyzed annulation with electron-deficient olefins to produce methylenecyclopentanes via the
trimethylenemethane–
palladium complex. This cycloaddition has served as a key step in synthetic approaches directed toward natural products such as brefeldin A
6 and albene,
7 and the polyquinanes.
8 Additions to heteroatom unsaturation can also be induced to occur to form
oxygen and
nitrogen heterocycles
9 as well as carbocycles.
10 It has also been employed to generate the TMM complex of transition metals other than
palladium.
11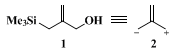
The present procedure provides a convenient two-step route to
2-(hydroxymethyl)allyltrimethylsilane using relatively inexpensive reagents. Other approaches require additional steps and expensive
chloromethyltrimethylsilane.
3,12Dianion formation from
2-methyl-2-propen-1-ol seems to be highly dependent on reaction conditions. Silylation of the dianion generated using a previously reported method
13 was unsuccessful in our hands. The procedure described here for the metalation of the
allylic alcohol is a modification of the one reported for formation of the dianion of
3-methyl-3-buten-1-ol.
14 The critical variant appears to be the polarity of the reaction medium. In solvents such as
ether and hexane, substantial amounts (15–50%) of the vinylsilane 3 are observed. Very poor yields of the desired product were obtained in
dimethoxyethane and
hexamethylphosphoric triamide, presumably because of the decomposition of these solvents under these conditions. Empirically, the optimal solvent seems to be a mixture of ether and
tetrahydrofuran in a ratio (v/v) varying from 1.4 to 2.2; in this case
3 becomes a very minor component.
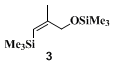
A similar procedure has been employed to silylate the dianion of
3-methyl-3-buten-2-ol (67% yield).
15 In systems where such internal activation is not possible (e.g.,
2-methyl-2-cyclohexen-1-ol), dianion formation can be performed in
hexane to give a 75% yield of the corresponding disilyl compound.
16
Appendix
Chemical Abstracts Nomenclature (Collective Index Number);
(Registry Number)
lithium alkoxide
sodium ketyl of benzophenone
sodiumketyl of benzophenone
ethanol (64-17-5)
potassium carbonate (584-08-7)
sulfuric acid (7664-93-9)
ether (60-29-7)
sodium bicarbonate (144-55-8)
allylic alcohol (107-18-6)
allyl (1981-80-2)
oxygen (7782-44-7)
nitrogen (7727-37-9)
copper sulfate (7758-98-7)
palladium,
palladium(0) (7440-05-3)
lithium (7439-93-2)
butane (106-97-8)
3-methyl-3-buten-2-ol (10473-14-0)
butyllithium (109-72-8)
Tetrahydrofuran (109-99-9)
2-methyl-2-propen-1-ol (513-42-8)
hexane (110-54-3)
calcium hydride (7789-78-8)
dimethoxyethane (534-15-6)
trimethylenemethane (287-23-0)
tributylamine (102-82-9)
hexamethylphosphoric triamide (680-31-9)
CHLOROTRIMETHYLSILANE (75-77-4)
hexamethyldisiloxane (107-46-0)
tetramethylethylenediamine (20485-44-3)
2-METHYL-2-PROPEN-1-OL DIANION
2-(Hydroxymethyl)allyltrimethylsilane (81302-80-9)
2-(trimethylsiloxy)allyltrimethylsilane
2-(Trimethylsiloxymethyl)allyltrimethylsilane
dibromoethane (557-91-5)
vinylsilane (7291-09-0)
2-methyl-1-trimethylsiloxy-3-trimethylsilyl-2-propene
2-(acetoxymethyl)allyltrimethylsilane (72047-94-0)
chloromethyltrimethylsilane (2344-80-1)
3-methyl-3-buten-1-ol (763-32-6)
2-methyl-2-cyclohexen-1-ol
Copyright © 1921-, Organic Syntheses, Inc. All Rights Reserved