Org. Synth. 2002, 78, 36
DOI: 10.15227/orgsyn.078.0036
INDOLE SYNTHESIS BY Pd-CATALYZED ANNULATION OF KETONES
WITH o-IODOANILINE: 1,2,3,4-TETRAHYDROCARBAZOLE
[
1H-Carbazole, 2,3,4,9-tetrahydro-
]
Submitted by Cheng-yi Chen and Robert D. Larsen
1
.
Checked by Adam Charnley and Steven Wolff.
1. Procedure
To a 100-mL, two-necked flask, is added a mixture
of
cyclohexanone (5.9 g,
60 mmol)
,
o-iodoaniline
(4.4 g, 20 mmol)
, and
1,4-diazabicyclo[2.2.2]octane
(DABCO) (6.7 g, 60 mmol)
in
N,N-dimethylformamide
(DMF) (60 mL). The mixture is degassed three times via
nitrogen/vacuum, followed by the addition of
palladium
acetate (Pd(OAc)2) (2.24 mg, 0.1 mmol)
(Note 1). The mixture is degassed twice and heated at 105°C for
3 hr or until completion of the reaction (Note 2). The reaction
mixture is cooled to room temperature and partitioned between
isopropyl
acetate (150 mL) and water (50 mL).
The organic layer is separated, washed with brine (50
mL), and concentrated under vacuum to dryness. The residue is
chromatographed on 50 g of silica gel using
700 mL
of ethyl acetate-heptane
(1:6)
as the eluent to give 2.22 g of
1,2,3,4-tetrahydrocarbazole
(65%) as a pale brown solid
(Note 3).
2. Notes
1.
Both
cyclohexanone
and o-iodoaniline were purchased from Lancaster Synthesis
and used directly in the reaction without further purification.
2.
The reaction generally takes 3-5 hr to complete and is monitored
by TLC (R
f = 0.50, SiO
2, eluted with
EtOAc
-
heptane, 1:4).
3.
The product is fully characterized:
mp 116-118°C; IR (neat)
cm
−1: 3401, 2928, 2848, 1470,
1305, 1235, 739
;
1H NMR (300 MHz, CDCl
3) δ:
1.86-1.99 (br m, 4 H), 2.74 (br t, 4 H, J = 6), 7.08-7.71
(br m, 2 H), 7.29 (m, 1 H), 7.49 (m, 1 H), 7.64
(br s, 1 H)
;
13C
NMR (75 MHz, CDCl
3) δ: 20.05, 22.20, 22.32,
22.42, 108.98, 109.61, 116.81,
118.12, 119.96, 126.82, 133.30,
134.66
. Anal. Calcd for C
12H
13N: C,
84.17; H, 7.65; N, 8.18. Found: C, 82.87; H, 7.53; N, 7.84.
Handling and Disposal of Hazardous Chemicals
The procedures in this article are intended for use only by persons with prior training in experimental organic chemistry. All hazardous materials should be handled using the standard procedures for work with chemicals described in references such as "Prudent Practices in the Laboratory" (The National Academies Press, Washington, D.C., 2011 www.nap.edu). All chemical waste should be disposed of in accordance with local regulations. For general guidelines for the management of chemical waste, see Chapter 8 of Prudent Practices.
These procedures must be conducted at one's own risk. Organic Syntheses, Inc., its Editors, and its Board of Directors do not warrant or guarantee the safety of individuals using these procedures and hereby disclaim any liability for any injuries or damages claimed to have resulted from or related in any way to the procedures herein.
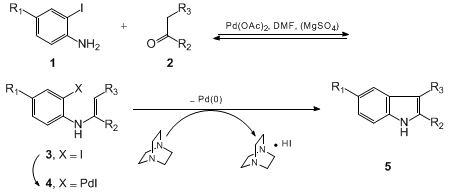
3. Discussion
The
indole nucleus is a common and important feature of a
variety of natural products and medicinal agents.
2 The traditional approach for
preparing the
indole nucleus is the Fischer indole reaction.
3 As this reaction has shortcomings,
the
palladium-catalyzed coupling of ortho-haloanilines is becoming
an excellent alternative.
4 Recently, the submitters disclosed a new and
efficient method for
indole synthesis using a
palladium-catalyzed annulation
between o-iodoanilines and ketones (Scheme 1).
5
As illustrated in Chart 1, this reaction is applicable to a variety of o-iodoanilines
and cyclic ketones to prepare the desired indoles in good yields. The coupling reaction
is highly regioselective.
6
For example, condensation of
o-iodoaniline
1, R=1-(1,2,4-triazolyl)methyl,
with
2-methylcyclohexanone gave
tetrahydrocarbazole
11 in
68% yield. Reaction
of
3-methylcyclohexanone with
o-iodoaniline
formed
tetrahydrocarbazole
12 predominantly. The reaction
is also compatible with
cyclopentanone and
cycloheptanone
(compounds
8 and
9). The reaction tolerates a variety of functional
groups, especially the acid-sensitive ketal (
10), carbamate (
14), or
the benzyl triazole
7
(
8,
9 and
11). These compounds, which would be unstable under
the conditions of the traditional Fischer indole reaction,
8 were conveniently synthesized using this
method. The structurally interesting
indole
15 was prepared
from
3-quinuclidinone hydrochloride
(1.0 equiv) in 55% yield. The interesting coupling of the
indole
nucleus onto a steroid was also achieved with
5α-cholestanone
(1.0 equiv) affording
13 exclusively in 79% yield.
Pyruvic
acid and
acetyl silane were also acceptable substrates,
used to prepare indoles
16 and
17 in 82% and 64% yield respectively.
Overall, the simple procedure, mild reaction conditions, and availability of the starting
materials render this method a valuable addition to
indole chemistry.
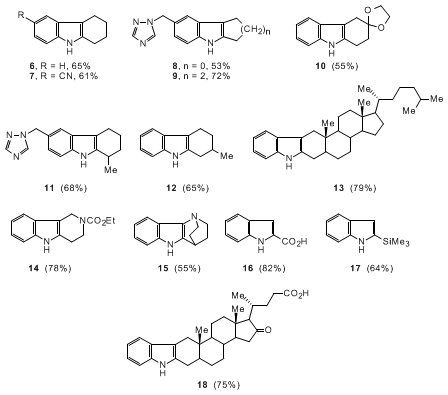
The high regioselectivity of these reactions follows the same pattern as those
of 2- and 3-substituted cyclohexanones when converted to enamines.
9 Apparently, A
1,2 and A
1,3
strain in the transition state controls the regiochemistry. The additive
magnesium
sulfate (MgSO
4), presumably acting as a dehydrating agent, was
found to promote the annulation (for compounds
7,
8,
11,
13,
15 and
17), indicating that the formation of the imine or enamine intermediate
is critical to the reaction. The annulation of
dehydrocholic acid
and
o-iodoaniline clearly demonstrated the high efficiency of
this reaction as both excellent chemoselectivity and regioselectivity were observed
(
18). The coupling reaction led to the unique combination of
indole
and steroid moieties into one interesting molecule.
Appendix
Chemical Abstracts Nomenclature (Collective Index Number);
(Registry Number)
o-Iodoaniline:
Aniline, o-iodo-
(8);
Benzenamine, 2-iodo- (9); (615-43-0)
1,2,3,4-Tetrahydrocarbazole:
Carbazole, 1,2,3,4-tetrahydro-
(8);
1H-Carbazole, 2,3,4,9-tetrahydro- (9); (942-01-8)
Cyclohexanone (8,9); (108-94-1)
1,4-Diazabicyclo[2.2.2]octane: DABCO (8,9); (280-57-9)
N,N-Dimethylformamide: CANCER SUSPECT
AGENT:
Formamide, N,N-dimethyl- (8,9); (68-12-2)
Palladium acetate:
Acetic acid, palladium(2+)
salt (8,9); (3375-31-3)
Copyright © 1921-, Organic Syntheses, Inc. All Rights Reserved