Org. Synth. 2005, 81, 171
DOI: 10.15227/orgsyn.081.0171
SYNTHESIS OF 9-SPIROEPOXY-endo-TRICYCLO[5.2.2.0]UNDECA-4,10-DIEN-8-ONE
[(Spiro[4,7-ethano-1H-indene-8,2'-oxiran]-9-one, 3a,4,7a-tetrahydro-)]
Submitted by Vishwakarma Singh*, Mini Porinchu, Punitha Vedantham, and Pramod K. Sahu
1.
Checked by Aaron Murray and Marvin J. Miller.
1. Procedure
9-Spiroepoxy-endo-tricyclo[5.2.2.02,6]undeca-4,10-dien-8-one. A 2-L, single-necked, round-bottomed flask equipped with a magnetic stirbar is charged with salicyl alcohol 1 (20.0 g, 161 mmol) (Note 1) and 325 mL of acetonitrile (Note 2). The resulting solution is cooled at 0°C in an ice bath while freshly generated cyclopentadiene (20 mL, 15.6 g, 236 mmol) (Note 3) is added in one portion. A pressure-equalizing addition funnel is then attached to the flask and charged with a solution of sodium metaperiodate (60 g, 280 mmol) (Note 4) in 220 mL of water. This solution is added dropwise to the ice-cooled, vigorously stirred reaction mixture over a period of 2 hr (Note 5). The color of the reaction mixture initially turns yellow-orange. After the reaction mixture is stirred at 0°C for an additional 1 hr, additional cyclopentadiene (10 mL) is added and stirring is continued for 30 min at 0°C. The ice bath is then removed and after 30 min an additional 10-mL portion of cyclopentadiene (Note 6) is added and the reaction mixture is stirred overnight at room temperature (Note 7).
Sodium chloride (50 g) is added to the reaction mixture and the suspension is stirred for 1 hr. The reaction mixture is then filtered under vacuum through a 30 × 35 mm pad of Celite in a sintered glass funnel washing with two 50-mL portions of diethyl ether. Two phases form in the filtrate. The organic phase is separated and concentrated by rotary evaporation. The residue is combined with the aqueous phase and extracted with four 100-mL portions of ether, and the combined organic phases are washed with 50 mL of brine, dried over anhydrous sodium sulfate, filtered, and concentrated to afford 32.5 g of an orange-brown oil which is adsorbed onto 25 g of silica gel and charged onto a 430 mm × 45 mm column of 350 g of 60-120 mesh silica gel. The column is eluted with ca. 700 mL of petroleum ether (bp 60-80°C) to remove hydrocarbon impurities, and elution is then continued with 500 mL of 98:2 petroleum ether and then with 95:5 petroleum ether:ethyl acetate to give a small amount of salicylaldehyde (Note 8). Further elution with 3.0 L of 95:5 petroleum ether:ethyl acetate gives pure 3. The column is further eluted with 90:10 petroleum ether-ethyl acetate to give 3.5 g of starting material (1). Recrystallization of 3 from a mixture of 25 mL of ether and 15 mL of petroleum ether gives 18.57-19.5 g (61-64%) of pure 3 as a white solid (Note 9).
2. Notes
1.
Salicyl alcohol was purchased from Lancaster, UK or from Fluka and used as received.
2.
The submitters purchased
acetonitrile (AR grade) from Sisco Research Laboratories, India and used it as received. The checkers obtained
acetonitrile from Aldrich Chemical Company.3.
Cyclopentadiene was obtained by pyrolysis of dicyclopentadiene (pract., 90%) which was purchased from Fluka. Pyrolysis was carried out following the previously described general procedure.
2,
Dicyclopentadiene (80 mL) was placed in a round-bottomed flask equipped with a
magnetic stirbar and a Vigreux column fitted with a distillation head through which cold water was circulated. The contents of the flask were slowly heated with stirring at 160°C in an
oil bath, and
ca. 60 mL of cyclopentadiene (bp 38-42°C) was collected in a receiver cooled in an ice-salt bath. The
cyclopentadiene was used immediately and the residue left in the distillation flask was discarded.
4.
Sodium metaperiodate was purchased by the submitters from S.D.Fine Chemicals, India and by the checkers from Aldrich Chemical Company. Occasionally a small amount of some material remains insoluble and is removed by filtration; the checkers observed that it begins to precipitate during the addition. An excess of the reagent was used so that oxidation is as complete as possible.
5.
A powerful
magnetic stirrer is required since a large amount of inorganic material precipitates after the addition of
sodium metaperiodate. The submitters used a
35-mm stirbar to achieve vigorous stirring.
6.
An excess of
cyclopentadiene was used because of its volatility and tendency to dimerize.
Cyclopentadiene was added intermittently in portions so that a sufficient amount is always present in the reaction mixture to efficiently intercept the
spiroepoxycyclohexadienone (
2) generated
in situ.
7.
Analysis by thin layer chromatography (TLC) indicates product formation and a small amount of starting material. TLC was performed on Merck Silica gel 60 F-
254 coated on aluminum sheets. Product
3 has R
f = 0.44 (elution with
70:30 petroleum ether-ethyl acetate; visualization with iodine vapor).
8.
Some
salicylaldehyde is also formed during the oxidation.
9.
The physical properties of
3 were as follows:
mp 40-41°C; IR (neat) cm
−1: 3058, 2931, 1734;
1H NMR
pdf (300 MHz, CDCI
3): δ 2.01 (m, 1 H), 2.57 (m, 1 H), 2.63 (m, 1 H), 2.81 (m, 1 H), 3.03 (m, 1 H), 3.08 (m, 1 H), 3.34 (m, 2 H), 5.44 (m, 1 H), 5.69 (m, 1 H), 6.10 (t, superimposed dd, J
1 = J
2 =7.2, 1 H), 6.38 (t, superimposed dd, J
1= J
2=7.2, 1 H);
13C NMR (75 MHz, CDCI
3) δ: 36.3, 38.5, 44.1, 50.4, 52.4, 52.8, 58.1, 126.4, 129.6, 132.2, 133.5, 205.3. These spectral features match data reported earlier.
4a The orientation of the oxirane ring was suggested on the basis of general tendency of cyclohexa-2,4-dienones during their cycloaddition and comparison with other similar adducts.
3,4
Handling and Disposal of Hazardous Chemicals
The procedures in this article are intended for use only by persons with prior training in experimental organic chemistry. All hazardous materials should be handled using the standard procedures for work with chemicals described in references such as "Prudent Practices in the Laboratory" (The National Academies Press, Washington, D.C., 2011 www.nap.edu). All chemical waste should be disposed of in accordance with local regulations. For general guidelines for the management of chemical waste, see Chapter 8 of Prudent Practices.
These procedures must be conducted at one's own risk. Organic Syntheses, Inc., its Editors, and its Board of Directors do not warrant or guarantee the safety of individuals using these procedures and hereby disclaim any liability for any injuries or damages claimed to have resulted from or related in any way to the procedures herein.
3. Discussion
This preparation is a modification of a general method developed in our laboratory for syntheses of a variety of tricycloundecadienones of type (
3) which involves
in-situ generation of spiroepoxycyclohexa-2,4-dienones and their interception with dienes.
3 This method is fairly versatile and a number of tricycloundecadienones have been prepared by oxidation of hydroxymethyl phenols and interception of the resulting cyclohexadienones with
cyclopentadiene,
spiro[4,2]cycloheptadiene and
dimethylfulvene.
4 This synthesis also constitutes an example of generation of molecular complexity in a single step from simple starting materials.
5 Recently, interception of cyclohexa-2,4-dienones with electron-rich olefins has also been reported.
6 Oxidation of hydroxymethyl phenols with
sodium metaperiodate has been extensively studied and is known to involve spiroepoxycyclohexa-2,4-dienones which are highly unstable and undergo dimerization instantaneously.
7 Recently, substituted spiroepoxycyclohexadienones were isolated and used as precursors in synthesis.
8,9 Oxidation of
5-bromosalicyl alcohol was also reported to give a stable
4-bromo-6-spiroepoxycyclohexa-2,4-dienone.
10Though the cycloaddition described here gives the adduct
3 in a regio- and stereoselective fashion, there exists a mechanistic dichotomy regarding the mode of the pericyclic reaction between cyclohexa-2,4-dienones with dienes.
11,12 The
spiroepoxycyclohexa-2,4-dienone 2 may participate as a 4-π component (diene) and cyclopentadiene as a 2-π partner (dienophile) to give the adduct
3 directly. Alternatively, the
cyclohexa-2,4-dienone may react as a 2-π partner (dienophile) and
cyclopentadiene as a 4-π component (diene) to give the adduct of type
I which may undergo a 3,3-shift
12 to give the product
3. However, we were unable to isolate adducts of type
I under our experimental conditions.
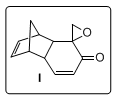
The epoxyketone
3 is a versatile precursor for a variety of tricycloundecane systems having a β,γ-unsaturated carbonyl chromophore, which are not so readily accessible. The contiguous epoxy ketone functionality and the double bond present in the five-membered ring provide opportunities for further manipulation. Adduct
3 may be transformed into a variety of molecular frameworks such as linearly fused
cis:anti:cis tricyclopentanoids, protoilludanes, and marasmanes in a stereoselective fashion after suitable chemical and photochemical manipulation.
13
Appendix
Chemical Abstracts Nomenclature (Collective Index Number);
(Registry Number)
9-Spiroepoxy-endo-tricyclo[5.2.2.02,6]undeca-4,10-dien-8-one:
Spiro[4,7-ethano-1H- indene-8,2'-oxiran]-9-one, 3a,4,7,1,7a-tetrahydro-; (146924-02-9)
Salicyl alcohol:
Benzenemethanol, 2-hydroxy-; (90-01-7)
Cyclopentadiene:
1,3-Cyclopentadiene; (542-92-7)
Sodium metaperiodate:
Periodic acid (HIO4), sodium salt; (7790-28-5)
Dicyclopentadiene:
4,7-Methano-1H-indene, 3a,4,7,7a-tetrahydro-; (77-73-6)
Copyright © 1921-, Organic Syntheses, Inc. All Rights Reserved